|
Please help improve this article by adding citations to reliable sources.Find sources: Biopharmaceutics Classification System news newspapers books scholar JSTOR ( July 2019 ) ( Learn how and when to remove this template message ).The solubility classification is based on a United States Pharmacopoeia (USP) aperture.The intestinal permeability classification is based on a comparison to the intravenous injection.

A correlation between the in vivo bioavailability and the in vitro solvation can be found. Usually they are not well absorbed over the intestinal mucosa and a high variability is expected. A drug is considered highly soluble when the highest dose strength is soluble in 250 ml or less of aqueous media over the pH range of 1 to 7.5. The volume estimate of 250 ml is derived from typical bioequivalence study protocols that prescribe administration of a drug product to fasting human volunteers with a glass of water. 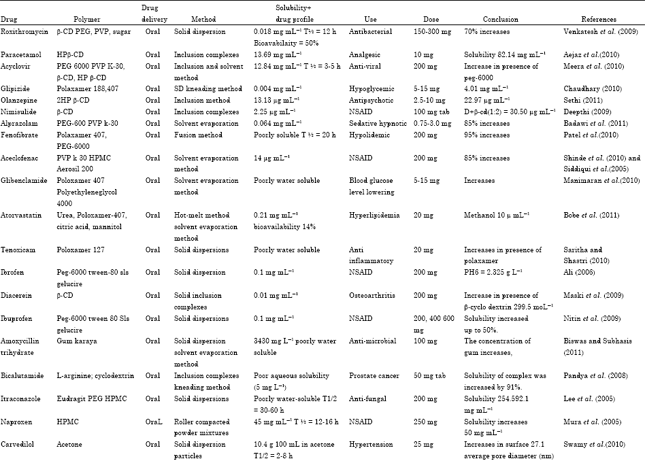
A drug substance is considered highly permeable when the extent of absorption in humans is determined to be 90 or more of the administered dose based on a mass-balance determination or in comparison to an intravenous dose. Drug Bioavailability: Estimation of Solubility, Permeability, Absorption and Bioavailability (Methods and Principles in Medicinal Chemistry). A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm. Res. 12 (3): 41320. By using this site, you agree to the Terms of Use and Privacy Policy. Wikipedia is a registered trademark of the Wikimedia Foundation, Inc., a non-profit organization.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Home
- About Brooke
- Blog
- Contact
- Nested for loop in r
- Realplayer 16 free download
- Tone2 electra 2 download
- Migrate nsf to pst
- Whitney houston lifetime movie online
- Motorola cricket 7
- Download lagu sekali ini saja gleen fredly
- Lg flatron e2350v screen goes to sleep
- Sap ecc 6-0 free trial download
- My hero academia movie 3
- Pioneer djm 2000 photo
- What is autodesk
- Ravenfield beta 7 download
 RSS Feed
RSS Feed
